Uncovering the Role of Cutaneous Microbiota in the Immunopathogenesis and Therapeutic Resistance of Merkel Cell Carcinoma: A Precision Oncology Perspective
Keywords:
Merkel cell carcinoma, cutaneous microbiota, Merkel cell polyomavirus, immune checkpoint inhibitors, precision oncology, microRNA biomarkersAbstract
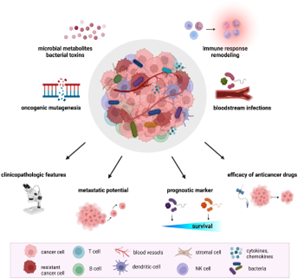
Merkel cell carcinoma (MCC) is a rare but highly aggressive cutaneous neuroendocrine malignancy predominantly affecting immunocompromised and elderly individuals. Driven by Merkel cell polyomavirus (MCPyV) in over 80% of Western cases and by ultraviolet (UV)-induced mutagenesis in the remainder, MCC presents significant clinical challenges due to propensity for rapid metastasis and resistance to conventional therapies. The cutaneous microbiota has recently emerged as a critical modulator of tumour immune microenvironment composition, antitumour surveillance, and responsiveness to immune checkpoint inhibitors (ICIs). This review synthesises current evidence on MCC epidemiology, viral oncogenesis, tumour microenvironment (TME) biology, and ICI therapy with evolving insights into host-microbiome interactions. We explore how microbial dysbiosis may potentiate immunopathogenesis and attenuate ICI efficacy, and we highlight translational opportunities including fecal microbiota transplantation (FMT), defined bacterial isolates, and bacterial consortia as microbiome-targeted adjuncts within a precision oncology framework.
Downloads
References
Toker C. Trabecular carcinoma of the skin. Arch Dermatol. 1972;105(1):107–10. https://doi.org/10.1001/archderm.1972.01620040075020
Tang CK, Toker C. Trabecular carcinoma of the skin: an ultrastructural study. Cancer. 1978;42(5):2311–21. https://doi.org/10.1002/1097-0142(197811)42:5<2311::AID-CNCR2820420531>3.0.CO;2-L
Feng H, Shuda M, Chang Y, Moore PS. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science. 2008;319(5866):1096–100. https://doi.org/10.1126/science.1152586
Heath M, Jaimes N, Lemos B, Mostaghimi A, Wang LC, Peñas PF, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients. J Am Acad Dermatol. 2008;58(3):375–81.
Paulson KG, Iyer JG, Blom A, Warton EM, Sokil M, Yelistratova L, et al. Systemic immune suppression predicts diminished Merkel cell carcinoma–specific survival. J Am Acad Dermatol. 2010;63(5):e81–7. https://doi.org/10.1016/j.jaad.2017.10.028
Becker JC. Merkel cell carcinoma. Ann Oncol. 2010;21(Suppl 7):vii81–5.
Allen PJ, Bowne WB, Jaques DP, Brennan MF, Busam K, Coit DG. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23(10):2300–9.
Harms PW, Harms KL, Moore PS, DeCaprio JA, Nghiem P, Wong MK, et al. The biology and treatment of Merkel cell carcinoma: current understanding and research priorities. J Invest Dermatol. 2018;138(2):e11–21. https://doi.org/10.1038/s41571-018-0103-2
Lemos BD, Nghiem P. Merkel cell carcinoma: more deaths but still no pathway to blame. J Invest Dermatol. 2007;127(9):2100–3.
Shuda M, Feng H, Kwun HJ, Rosen ST, Gjoerup O, Moore PS, et al. T antigen mutations are a human tumor-specific signature for Merkel cell polyomavirus. Proc Natl Acad Sci U S A. 2008;105(42):16272–7.
Harms PW, Vats P, Verhaegen ME, Robinson DR, Wu YM, Dhanasekaran SM, et al. The distinctive mutational spectra of polyomavirus-negative Merkel cell carcinoma. Cancer Res. 2015;75(18):3720–7.
Iyer JG, Afanasiev OK, McClurkan C, Paulson K, Nagase K, Jing L, et al. Merkel cell carcinoma in immunosuppressed patients: clinical features and outcomes. J Am Acad Dermatol. 2014;70(4):684–92.
Sunshine JC, Jahchan NS, Sage J, Choi J. Are there multiple cells of origin of Merkel cell carcinoma? Oncogene. 2018;37(11):1409–16. https://doi.org/10.1038/s41388-017-0024-6
Paulson KG, Carter JJ, Johnson LG, Cahill KW, Iyer JG, Schrama D, et al. Antibodies to Merkel cell polyomavirus T antigen oncoproteins reflect tumor burden in Merkel cell carcinoma patients. Cancer Res. 2010;70(21):8388–97. https://doi.org/10.1158/0008-5472.CAN-10-2128
Sihto H, Böhling T, Kavola H, Koljonen V, Salmi M, Jalkanen S, et al. Tumor infiltrating immune cells and outcome of Merkel cell carcinoma: a population-based study. Clin Cancer Res. 2012;18(10):2872–81. https://doi.org/10.1158/1078-0432.CCR-11-3020
Inoue T, Yoneda K, Manabe M, Demitsu T. Spontaneous regression of Merkel cell carcinoma: a case report and review of the literature. J Dermatol. 2012;39(3):273–6. https://doi.org/10.1111/j.1346-8138.2011.01363.x
Belkaid Y, Segre JA. Dialogue between skin microbiota and immunity. Science. 2014;346(6212):954–9. https://doi.org/10.1126/science.1260144
Lai Y, Cogen AL, Radek KA, Park HJ, Macleod DT, Leichtle A, et al. Activation of TLR2 by Staphylococcus epidermidis induces antimicrobial peptide expression. J Immunol. 2010;184(10):5427–36. https://doi.org/10.4049/jimmunol.0903902
Scharschmidt TC, Vasquez KS, Truong HA, Gearty SV, Pauli ML, Nosbaum A, et al. A wave of regulatory T cells into neonatal skin mediates tolerance to commensal microbes. Immunity. 2015;43(5):1011–21. https://doi.org/10.1016/j.immuni.2015.10.016
Cogen AL, Nizet V, Gallo RL. Skin microbiota: a source of disease or defence? Br J Dermatol. 2008;158(3):442–55. https://doi.org/10.1111/j.1365-2133.2008.08437.x
Naik S, Bouladoux N, Linehan JL, Han SJ, Harrison OJ, Wilhelm C, et al. Commensal-dendritic-cell interaction specifies a unique protective skin immune signature. Nature. 2015;520(7545):104–8. https://doi.org/10.1038/nature14052
Paulson KG, Iyer JG, Tegeder AR, Thibodeau R, Schelter J, Koba S, et al. Transcriptome-wide studies of Merkel cell carcinoma reveal immune signatures associated with survival. J Invest Dermatol. 2011;131(9):1813–21.
Afanasiev OK, Yelistratova L, Miller N, Nagase K, Paulson K, Iyer JG, et al. Merkel polyomavirus-specific T cells fluctuate with MCC burden. Cancer Immunol Res. 2013;1(1):27–37.
Nghiem PT, Bhatia S, Lipson EJ, Kudchadkar RR, Miller NJ, Annamalai L, et al. PD-1 blockade with pembrolizumab in advanced Merkel cell carcinoma. N Engl J Med. 2016;374(26):2542–52. https://doi.org/10.1056/NEJMoa1603702
Garrett WS. Cancer and the microbiota. Science. 2015;348(6230):80–6. https://doi.org/10.1126/science.aaa4972
Belkaid Y, Hand TW. Role of the microbiota in immunity and inflammation. Cell. 2014;157(1):121–41. https://doi.org/10.1016/j.cell.2014.03.011
Schwabe RF, Jobin C. The microbiome and cancer. Nat Rev Cancer. 2013;13(11):800–12. https://doi.org/10.1038/nrc3610
Gaiser MR, Bongiorno M, Brownell I. PD-1 blockade with pembrolizumab for advanced Merkel cell carcinoma: immune correlates and outcomes. J Immunother Cancer. 2016;4:99. https://doi.org/10.1186/s40425-016-0190-1
Schmidt BL, Kuczynski J, Bhattacharya A, Huey B, Corby PM, Queiroz EL, et al. Changes in abundance of oral microbiota associated with oral cancer. PLoS One. 2014;9(6):e98741. https://doi.org/10.1371/journal.pone.0098741
Starrett GJ, Thakuria M, Chen T, Marcelus C, Cheng J, Nomburg J, et al. Clinical and molecular characterization of virus-positive and virus-negative Merkel cell carcinoma. Genome Med. 2020;12(1):30. https://doi.org/10.1186/s13073-020-00727-4
Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19(11):1423–37. https://doi.org/10.1038/nm.3394
Pickup MW, Mouw JK, Weaver VM. The extracellular matrix modulates the hallmarks of cancer. EMBO Rep. 2014;15(12):1243–53. https://doi.org/10.15252/embr.201439246
Kalluri R. The biology and function of fibroblasts in cancer. Nat Rev Cancer. 2016;16(9):582–98. https://doi.org/10.1038/nrc.2016.73
Gascard P, Tlsty TD. Carcinoma-associated fibroblasts: orchestrating the composition of malignancy. Genes Dev. 2016;30(9):1002–19. https://doi.org/10.1101/gad.279737.116
Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005;307(5706):58–62. https://doi.org/10.1126/science.1104819
Carmeliet P, Jain RK. Angiogenesis in cancer and other diseases. Nature. 2000;407(6801):249–57. https://doi.org/10.1038/35025220
Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977. https://doi.org/10.1126/science.aau6977
Tkach M, Théry C. Communication by extracellular vesicles: where we are and where we need to go. Cell. 2016;164(6):1226–32. https://doi.org/10.1016/j.cell.2016.01.043
Zitvogel L, Ma Y, Raoult D, Kroemer G, Gajewski TF. The microbiome in cancer immunotherapy: diagnostic tools and therapeutic strategies. Science. 2018;359(6382):1366–70. https://doi.org/10.1126/science.aar6918
Hayes J, Peruzzi PP, Lawler S. MicroRNAs in cancer: biomarkers, functions and therapy. Trends Mol Med. 2014;20(8):460–9. https://doi.org/10.1016/j.molmed.2014.06.005
Renwick N, Cekan P, Masry PA, McGeary SE, Miller JB, Hafner M, et al. Multicolor microRNA FISH effectively differentiates tumor types. J Clin Invest. 2013;123(6):2694–702. https://doi.org/10.1172/JCI68779
Veija T, Sahi H, Koljonen V, Böhling T, Knuutila S, Sarhadi VK. MicroRNA expression profiling of Merkel cell carcinoma. Virchows Arch. 2015;467(5):561–70. https://doi.org/10.1007/s00428-015-1830-0
Routy B, Le Chatelier E, Derosa L, Duong CPM, Alou MT, Daillère R, et al. Gut microbiome influences efficacy of PD-1–based immunotherapy against epithelial tumors. Science. 2018;359(6371):91–7. https://doi.org/10.1126/science.aan3706
Sivan A, Corrales L, Hubert N, Williams JB, Aquino-Michaels K, Earley ZM, et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti–PD-L1 efficacy. Science. 2015;350(6264):1084–9. https://doi.org/10.1126/science.aac4255
Honda K, Littman DR. The microbiota in adaptive immune homeostasis and disease. Nature. 2016;535(7610):75–84. https://doi.org/10.1038/nature18848
Spencer CN, McQuade JL, Gopalakrishnan V, McCulloch JA, Vetizou M, Cogdill AP, et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science. 2021;374(6575):1632–40. https://doi.org/10.1126/science.aaz7015
Harshini K, Sathya A, Swathi G, Bhuvanesh M, Mariyammal R, Nepolean R. Fecal microbiota transplantation as a novel approach for Clostridium difficile infection: a review. GSC Biol Pharm Sci. 2025;31(3):351–8. https://doi.org/10.30574/gscbps.2025.31.3.0248
Published
Issue
Section
Categories
License
Copyright (c) 2026 Sathya Annadurai, R. Mariyammal, S.Vinothkumar, V. Elavarasan, M. Bhuvanesh, R.Napolean (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
License Terms
This is an open-access article published in the International Journal of Research in Pharmaceutical Sciences and Technology (IJRPST) by Rubatosis Publications.
It is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, reproduction, and adaptation in any medium or format, provided the original author(s) and source are appropriately credited, a link to the license is provided, and any changes made are indicated.
To view a copy of this license, visit: https://creativecommons.org/licenses/by/4.0/
For any further queries or permissions beyond the scope of this license, please contact: journals@rubatosis.org




